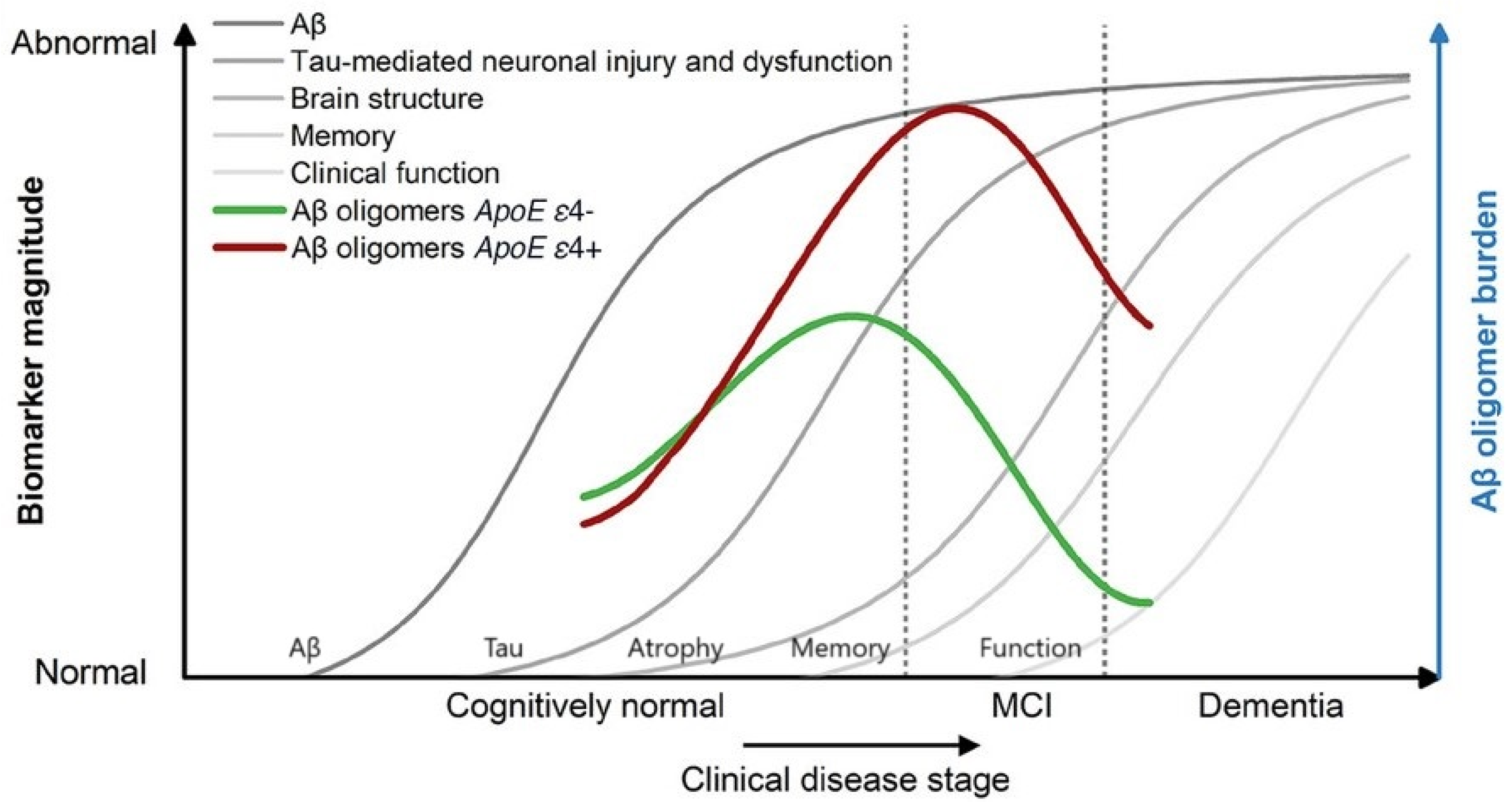
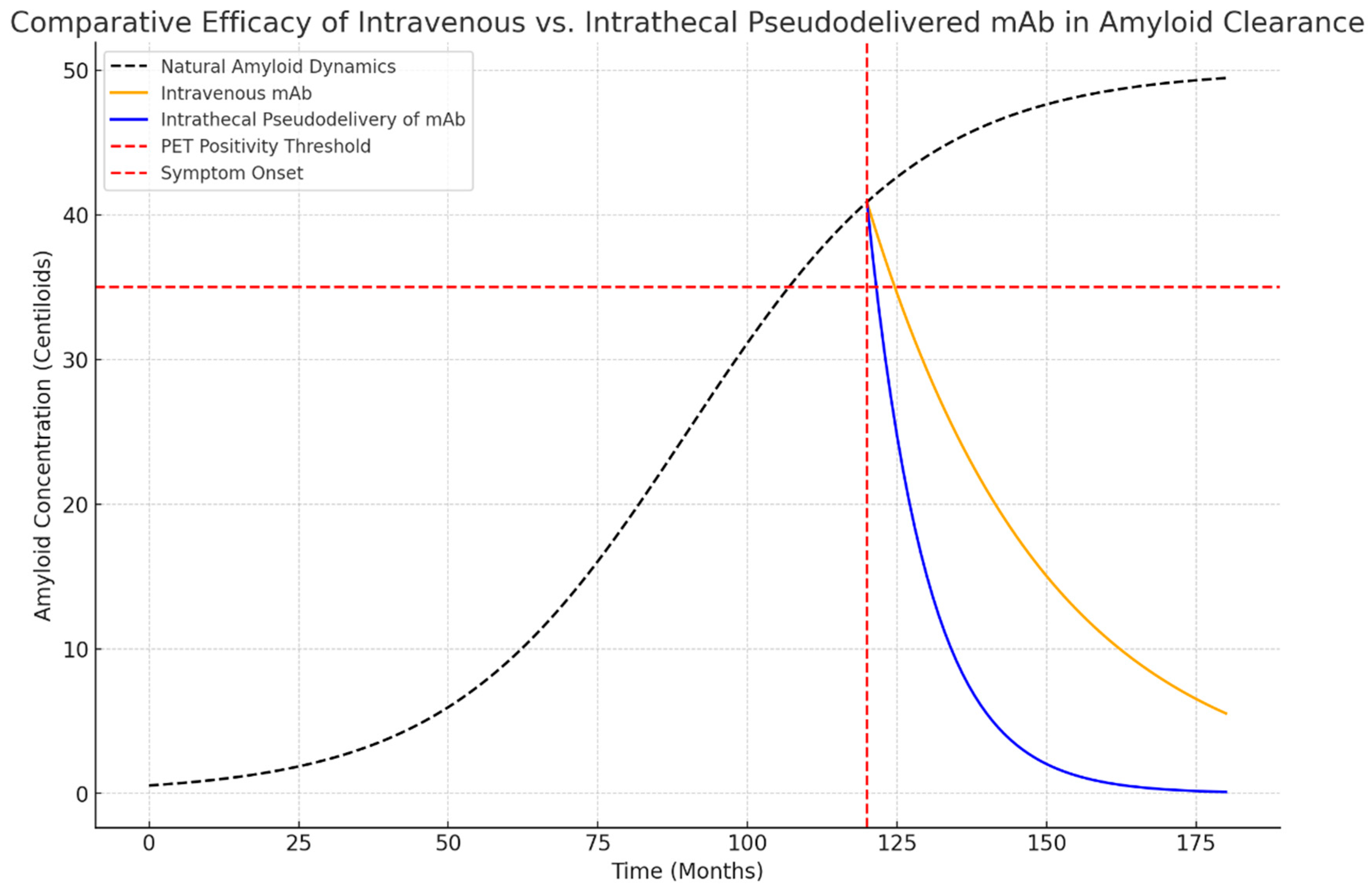
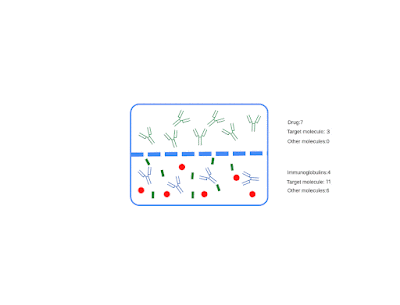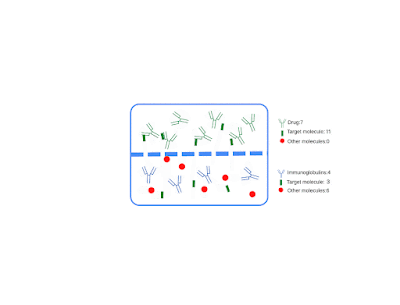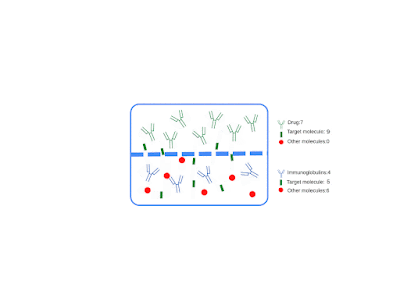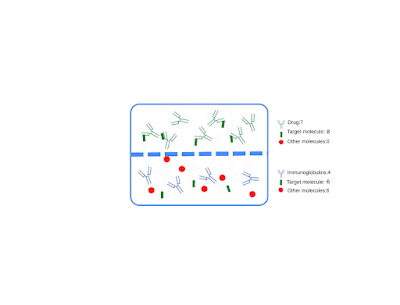
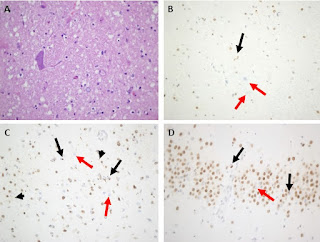
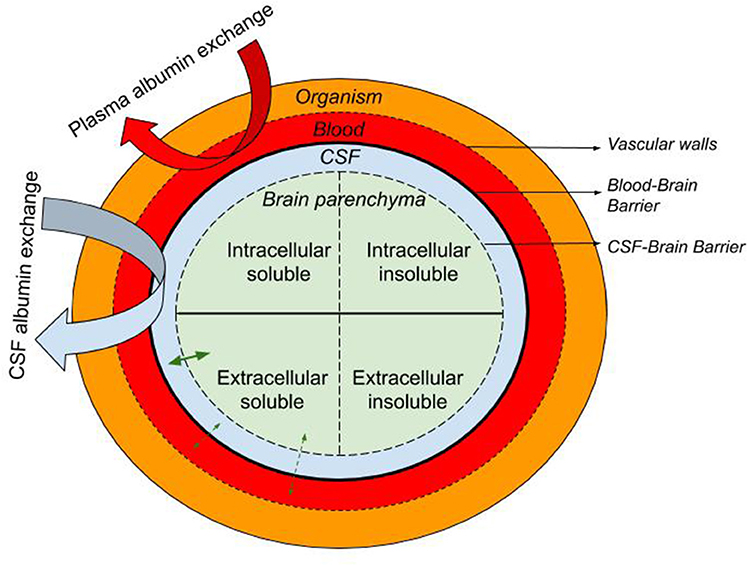
I am pleased to announce that I am editing a new volume titled "Intrathecal Drug Delivery", which will be published as part of the NEUROMETHODS series by Springer Nature. This book aims to provide a comprehensive and cutting-edge overview of intrathecal drug delivery, covering clinical applications, technological advancements, and experimental approaches.
We are currently assembling a diverse group of international experts in neurology, neurosurgery, anesthesiology, biomedical engineering, and related fields to contribute chapters to this volume. If you or a colleague are working on intrathecal drug delivery and would like to contribute, we invite you to express your interest!
Topics Already Confirmed in the Book Include:
✅ Existing and upcoming intrathecal drugs
✅ Parameters influencing drug distribution in the CSF
✅ Continuous and controlled intrathecal drug delivery
✅ Intrathecal drug delivery for chronic pain
✅ Investigating intrathecal treatment of leptomeningeal disease
✅ Pumpless implantable devices for intrathecal pseudodelivery of drugs
... and more!
Who Should Contribute?
We are looking for researchers, clinicians, and industry professionals with expertise in:
🔹 Neurology and Neurosurgery
🔹 Pain Medicine and Anesthesiology
🔹 Biomedical Engineering and Drug Delivery Systems
🔹 Neuropharmacology and Experimental Models of Intrathecal Therapies
Key Information for Authors:
📌 Manuscript submission deadline: May 31, 2025
📌 Chapter length: ~30–60 pages
📌 No cost for authors – Springer covers all publishing expenses
📌 Figures (including color images) are free of charge
📌 Global visibility – Springer Nature ensures high discoverability
If you are interested in contributing or know someone who might be a good fit, please reach out to me! This is a great opportunity to showcase your work in an internationally recognized book series.
Let’s shape the future of intrathecal drug delivery research together! 🚀
For inquiries or to propose a chapter, contact me at: menendezgmanuel @ uniovi.es
📢 Please share this post with colleagues who might be interested!
#IntrathecalDrugDelivery #Neuropharmacology #Neurosurgery #Neurology #PainMedicine #BiomedicalEngineering #DrugDelivery #SpringerNature #NEUROMETHODS #MedicalResearch